DFC Studies
Master Protocol: Biomarker Platform Study
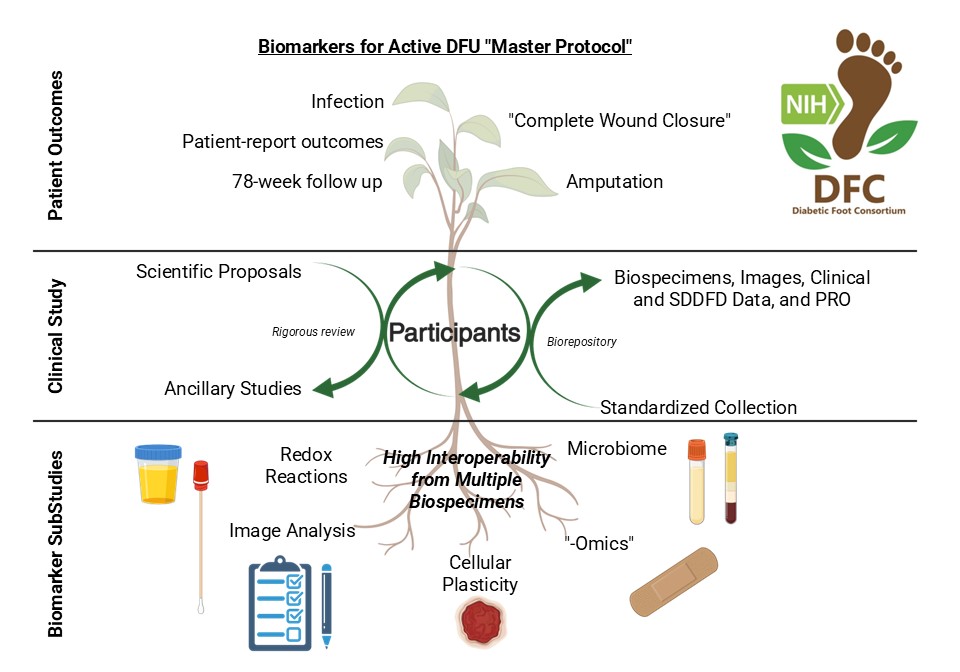
Open DFU Master Protocol
- Platform study designed to test multiple biomarkers to identify diabetic foot ulcers with higher healing potential.
- Participants enrolled from outpatient diabetic foot and wound care clinics across multiple healthcare systems.
- SABER serves as the DCC providing project management, data management, software development, and statistical analysis.
- Status: First subject enrolled June 29, 2023. Enrollment and follow-up ongoing.
- Ancillary applications encouraged. Email: DFC-Ancillary@umich.edu
- Please visit clinicaltrials.gov for more information.
CMYC
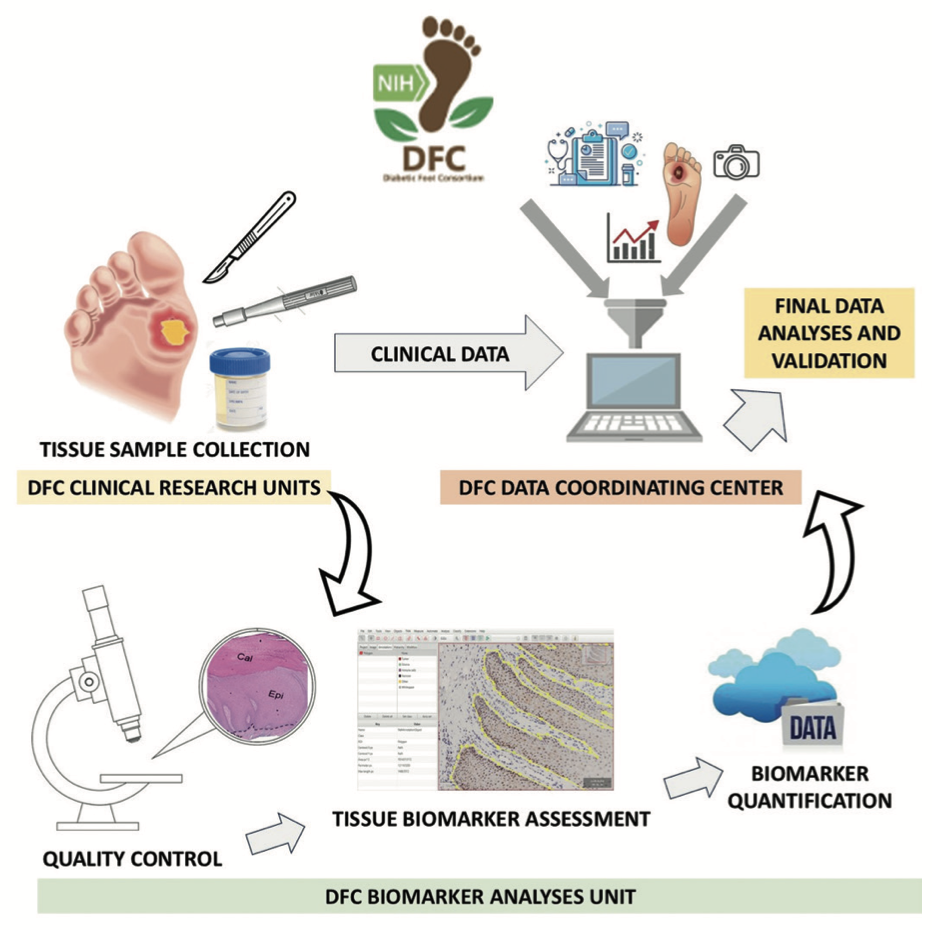
Predictive and Diagnostic Biomarkers for DFU – c-myc and p-GR
Multi-center observational cohort study to develop and validate tissue-based biomarkers predicting complete wound healing.
Tissue specimens collected at baseline and 4 weeks and analyzed centrally. Participants followed weekly for up to 12 weeks.
Please visit clinicaltrials.gov for more information.
Related Publications
TEWL
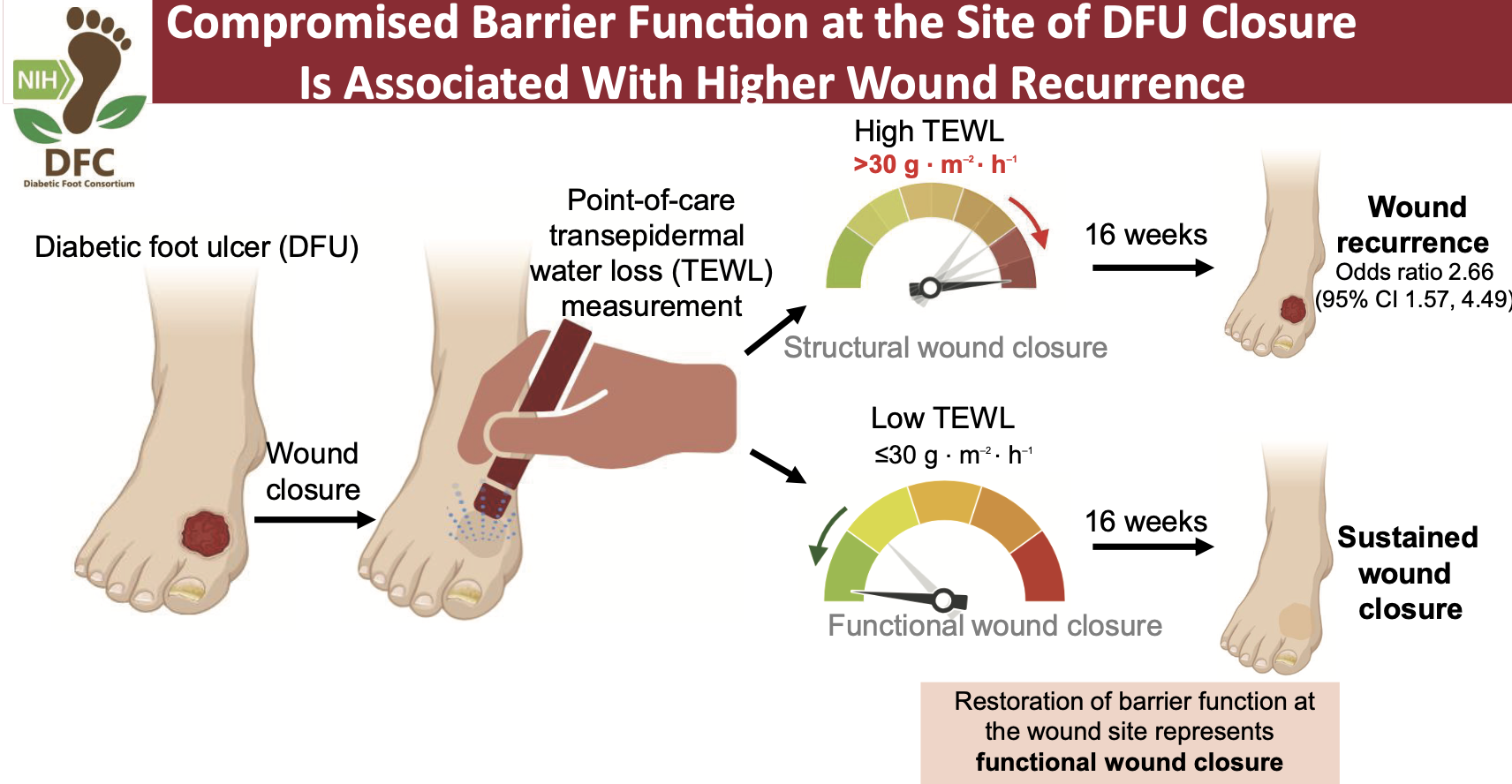
Trans-Epidermal Water Loss (TEWL) as a predictor of DFU recurrence
Multicenter study evaluating TEWL measurements as a predictor of DFU recurrence after wound closure.
TEWL measured at healed wound and reference sites. Participants followed weekly for 16 weeks or until recurrence.
Please visit clinicaltrials.gov for more information.
Biorepository
Adjunct collection of biorepository data from DFC trials
Standardized biorepository for biospecimen and clinical data collection across DFC trials.
Biospecimens and de-identified data archived in the NIDDK Central Repository for future research.
Study completed April 2024.
Please visit clinicaltrials.gov for more information.
Related publications
Projection of the 2050 diabetes burden in the US.